The Impact of Not Washing Hands on the Result of Capillary Glycemia
Josivan Lima, Deciara Mesquita, Kennyo Santos, Kallil Fernandes, Rodolpho Mendonca, Clementino Quinto, Dayse Silva, Lucia Helena Nobrega
1Department of Endocrinology, Onofre Lopes University Hospital/UFRN, Rua General Gustavo de Farias, S/N - Petrópolis, 59012-570, Natal, Rio Grande do Norte, Brazil
2Centro de Endocrinologia de Natal, Av, Afonso Pena, 757, 59020-100, Natal, Rio Grande do Norte, Brazil
- *Corresponding Author:
- Josivan Lima, 233, Natal-RN, Brazil
Tel: +5584-99878765
Fax +5584 32216324
E-mail: josivanlima@gmail.com
Received Date: November 12, 2015 Accepted Date: December 28, 2015 Published Date: January 05, 2016
Citation: Josivan Lima, Mesquita D, Santos K et al. (2016) The Impact of Not Washing Hands on the Result of Capillary Glycemia. J Clin Mol Endocrinol 1:2. doi: 10.21767/2572-5432.100002
Copyright: © 2016 Josivan Lima, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Introduction: The aim of this study is to quantify the influence of handling food on the results of capillary blood glucose (CBG), as well as evaluating whether using the second drop of blood could be more accurate.
Materials and methods: A total of 420 measurements of CBG was made in the following situations: hands not washed; hands washed; excess of alcohol on fingers; after handling orange, apple, strawberry, pineapple, banana, chocolate or bread with butter. A second measurement was performed after wiping the first drop and assessing a second. CBG after washing hands was considered as control. Fasting was not required.
Results: The mean glucose of the control group was 85.6±16.3 mg/dl. All groups showed higher results than the control (p<0.05), except for the group “bread with butter.” The mean levels for the 1st and 2nd drops were 106.6±40.9 mg/dl and 95.0±18.4 mg/dl, respectively. The difference between them was 11.4±3.2 mg/dl (p<0.001), and the difference between them and the control was +21.3±9.0 mg/dl (p=0.01) for the first drop and +8.1 ± 4.1 (p=0.05) for the second drop.
Discussion: There is a significant increase in the CBG measurements when the finger is exposed to food, even if it was extensively wiped. For this reason, patients should be advised to wash their hands with soap and water before performing the test. If that is not possible, patients must use the second drop of blood to minimize the increment.
Keywords
Capillary glycemia; Fingerstick glucose; Asepsis; Washing hands; Diabetes; Diabetic care
Introduction
The self-monitoring of blood glucose (SMBG) through capillary blood glucose (CBG) measurements, commonly called “finger-stick glucose,” is an important part of the management of diabetic patients [1]. The SMBG is currently recommended for patients with type 1 diabetes mellitus and many with type 2 diabetes mellitus, insulin treated or not [1-3].
However, this kind of assay can be biased by several factors, such as the innate variation error of the glucometer; not washing the hands properly before the sting; applying an inappropriate high pressure on the finger while milking it; having dirt on the meter; not covering all the target area of the strip; not obtaining an adequate blood drop; not starting the timer according to manufacturer’s guidelines; applying the blood incorrectly; having excess of alcohol (not dried) on the finger; inserting the strip incorrectly; and using expired strips [4].
To minimize such bias, organizations such as the Diabetes UK and the ADA recommend the measurement should be performed through a drop of blood taken from a finger after washing hands preferably with water and soap instead of wet wipes [5,6]. Despite this, one study showed that only 50% of patients wash their hands before testing [4].
When it is not possible to wash the hands properly before the SMBG, the Dutch Diabetes Association recommends that individuals use the second drop of blood after neglecting the first one with a handkerchief or napkin [7].
Our study has the objective of simulating daily life situations (handling food) and quantifying the influence of not washing the hands on the results of the capillary blood glucose in these situations. We also intend to determine whether using the second drop of blood would be a good alternative to minimize the effects of not washing hands.
Materials and Methods
We made a total of 420 measurements of CBG in different situations. These measurements were performed in 21 volunteers, 1 with type 2 diabetes and 20 without diabetes. The mean age was 31.4 ± 13.6 years old, and 13 (61.9%) were women. Fasting was not required.
Given the fact there is no difference between the baseline CBG on each finger [8], it was measured ten times for each patient, once on each finger as follows: 1) not washing the hands and before manipulating food; 2) after washing the hands with water and soap and drying them as the guidelines suggest [5,6]; 3) after putting excess alcohol on the finger and not waiting for it to dry; 4) after handling banana; 5) after handling orange; 6) after handling apple; 7) after handling strawberry; 8) after handling pineapple; 9) after handling chocolate and 10) after handling bread with a thin layer of butter. For each finger, an assistant would prick the volunteer’s distal phalanges with a lancet, milk it to access the first drop of blood, and then apply it on the glucometer. Afterward, the assistant would wipe the drop (only once) with sterile cotton and milk the finger again to obtain a second drop of blood and apply it on the glucometer.
The volunteers would perform the first monitoring as they walked in, before washing their hands or handling any of the foods. Afterward, they would wash their hands with soap and water and perform the measurement on the next finger. Then they would soak the third finger with alcohol and repeat the procedure before it dried.
For the remaining fingers, the volunteer had to touch and apply moderate pressure on each food in order for the distal phalanges to be visually wet or dirty; after that, the assistant would wipe the volunteer’s finger with a dry napkin until there was no visual detritus and then perform the new measurement. This procedure was performed one finger at a time, and the ten measurements for each patient were taken in no more than 20 minutes. During the experiment, no finger could touch the other.
The fruits were peeled and cut into small pieces. Sugared milk chocolate and full-fat salted butter were used in the chocolate and butter groups, respectively. The result of the CBG from the first drop of blood after washing and drying the hands was considered as a control for comparison purposes.
The International Organization for Standardization (ISO) guideline 15197:2013 suggests that for glucose levels below 100 mg/dl, the meter should read within 15 mg/dl of the reference sample, and the acceptable bias from the reference value should be within 15%. We used the same glucometer device for all measurements (Contour TS®). For this meter, about 90% of the measurements below 100 mg/dL have an innate measurement bias of less than 15%. To neutralize the bias of the innate variation of the glucometer, we compared the percentage of measurements that diverged more than 15% from the control in the first and second drops of blood.
Data is presented as mean ± SD. We obtained the difference between the means of the first and second drops and also between each of them and the control sample through a paired two-tailed t test. We also assessed the difference between each finger through ANOVA with the Bonferroni's multiple comparison post-test. A p value lower than 0.05 was considered statistically significant. The software used for the analysis was Graph pad Prism 5.0.
Results
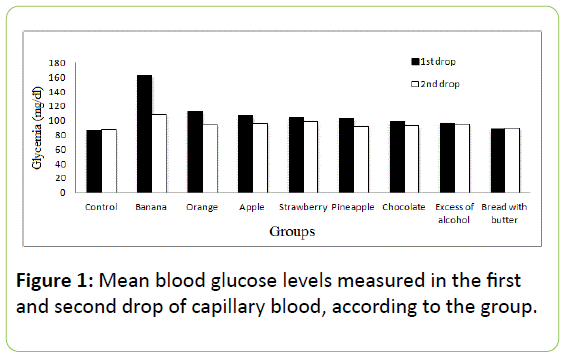
The mean glucose levels in the control group were 85.7 ± 16.3 mg/dl for the first drop and 87.4 ± 16.2 mg/dl for the second drop (Figure 1).
There was no statistically significant difference between them in this group (1.8 mg/dL; p=0.3). The mean glucose levels considering all the other groups together were 107.0 ± 40.8 mg/dl for the first and 95.5 ± 18.1 mg/dl for the second drop. We made two comparisons: first or second drop vs. control and first vs. second drop.
Compared with the control, there was a statistically significant increase of +21.3 ± 9 (p=0.01) in the first drop and a non-statistically significant increase of +8.1 ± 4.1 mg/dl (p=0.05) in the second drop. All groups showed a statistically significant higher first drop glycemia than the control sample (p <0.05), except for the group “bread with butter.” As for the second drop, there was no statistical difference in comparison with the control, except for the apple group (p=0.04), which still had higher glycemia measured in this drop.
Compared with the control, the average increase in CBG measured in the first drop (21.3 mg/dl) was greater than the 15% allowed by ISO 15197:2013, confirming the need to wash the hands before performing the test. About 50.3% of the results using the first drop diverged more than 15% compared with the control. Using the second drop, 30.9% diverged more than 15%.
There was a decrease of 11.4 ± 3.2 mg/dl (p<0.001) in glycemia measured in the second drop compared with the first drop. The following groups had no difference between the glycemia measured in the first and second drops (p>0.05): hands washed, strawberry, excess alcohol, bread with butter, and hands not washed.
Handling banana, orange, and apple caused the largest increases in glycemia. The banana group had the highest glycemic value, almost twice the control value (163 mg/dl). The mean results for each group, as well as the difference between the means compared with the control group, are listed in Table 1.
| Glycemia compared with control (mean±SD) | Increase in Glucose readings * | |||||||
|---|---|---|---|---|---|---|---|---|
| Group | 1st drop | p | 2nd drop | p | 1st drop | p | 2nd drop | p |
| Washing hands | 85.7 ± 16.3 | - | 87.4 ± 16.2 | >0.10 | 0 | - | 0 | - |
| Banana | 163 ± 81.1 | 0.018 | 109± 27.6 | 0.068 | +77.2 ± 76.6 | <0.001 | +21.6± 30.5 | 0.003 |
| Orange | 113.4 ± 93.5 | 0.065 | 93.5 ± 20.4 | >0.10 | +27.7 ± 36.1 | 0.002 | +6.1 ± 11 | 0.018 |
| Apple | 107.1 ± 9.7 | 0.034 | 96.4 ± 17.5 | 0.0411 | +21.4 ± 21.5 | <0.001 | +8.9 ± 18.9 | 0.041 |
| Strawberry | 104.2 ± 23 | >0.10 | 99.1 ± 13.4 | >0.10 | +18.5 ± 12.2 | <0.001 | +11.7 ± 11.1 | <0.001 |
| Pineapple | 102.7 ± 19 | >0.10 | 92.1 ± 13.9 | >0.10 | +17.0 ± 12.6 | <0.001 | +4.7 ± 13.1 | 0.106 |
| Chocolate | 98.7 ± 16.4 | >0.10 | 92.4 ± 15 | >0.10 | +13 ± 7.3 | <0.001 | +5.0 ± 11.6 | 0.060 |
| Excess of alcohol | 96.8 ± 27.87 | >0.10 | 95.3 ± 16.7 | >0.10 | +11.1 ± 18.5 | 0.012 | +7.9 ± 12.3 | 0.007 |
| Bread with butter | 88.2 ± 13.7 | >0.10 | 89.8 ± 14.6 | >0.10 | +2.5 ± 7 | 0.112 | +2.4 ± 7.4 | 0.181 |
| Fingers not washed | 88.9 ± 17.3 | 0.037 | 92.2 ± 14.3 | >0.10 | +3.3 ± 9.1 | 0.114 | +4.8 ± 9.7 | 0.035 |
| Total | 107.0 ± 0.8 | <0.001 | 95.5 ± 18.1 | <0.001 | +21.3 ± 9 | 0.018 | +8.1 ± 4.1 | 0.050 |
Table 1: Mean glycemic results for each group compared with control, as well as the difference between the means glucose readings in each drop sample
The differences between the results of the second drop and the first drop in each group are listed in Table 2.
| Mean±SD (mg/dl) | p * | |
|---|---|---|
| Washing hands | 1.8±7.9 | 0.318 |
| Banana | -53.9±78.7 | 0.005 |
| Orange | -19.9±31.6 | 0.009 |
| Apple | -10.7±27.8 | 0.091 |
| Strawberry | -5.1±20.3 | 0.263 |
| Pineapple | -10.5±14.4 | 0.003 |
| Chocolate | -6.3±1.2 | 0.018 |
| Excess of alcohol | -1.5±19 | 0.724 |
| Bread with butter | 1.6±5 | 0.151 |
| Hands not washed | 3.2±8.6 | 0.101 |
| Total | -11.4±3.2 | <0.001 |
Table 2: Difference (second measurement – first measurement) in glycemia readings using the second drop (comparing with the first drop).
Discussion
The measurement of the capillary glucose is a non-accurate method per se, and it can be even more deceiving when the patient performs it with dirty hands [4,8,9].
In this study, all the glycemia measurements made on dirty fingers were higher than those for the control, and the banana group had the highest level. Perhaps this could be due to the banana’s having a high concentration of glucose, compared with the other foods studied. We found that the capillary blood glucose could increase significantly if the finger was just submitted to contact with fruits or chocolate, even when the finger was extensively wiped until there was no visual detritus.
The bread with butter group was different from all other groups, and some patients had CBG values equal or even lower than the control group after handling the bread with butter. Besides the fact that the bread does not leave debris on the fingers, maybe the fat on the surface of the fingers after handling the bread and butter prevents the elevation in blood glucose. Likewise, the chocolate group had only a slight increase in glycemia, perhaps also due to the fat in chocolate. Maybe different chocolates can have different results.
All patients should be advised to wash their hands with water and soap as the guidelines for the SMBG recommend [5,6,9]. We did not find a statistically significant change between the baseline finger-stick (control) and the second drop of blood after handling food. For this reason, in situations where it is not possible to wash the hands, it could be acceptable to perform the measurement using the second drop of blood, after smoothly wiping away the first one.
Limitations of this study include the fact that our sample was not large, and the volunteers were mostly non-diabetics. However, our data are in agreement with some already described in the literature, and therefore, we do not think these limitations have changed our results.
Hortensius et al. [8] found a similar outcome. In their study, 88% of the volunteers had over 10% increase in glycemia compared with the control sample in the first drop of blood after handling fruits. However, only 11% of the volunteers persisted with this 10% increase in glycemia after wiping the finger and performing the second drop. Hirose et. al also found similar results of pseudohyperglycemia after fruit peeling and recommend to wash the hands before monitoring capillary blood glucose [10].
The internationally accepted guidelines for SMBG such as those provided by the ADA and the Diabetes UK do not mention the possibility of using the second drop of blood, but in the Netherlands, some guidelines present that recommendation as an alternative when the patients did not wash their hands [7]. If the hands have been adequately cleaned, there is no reason to perform the measurement in the second drop of blood, as there is no statistical difference between both results. Performing the second drop of blood could be an unnecessary distress for patients in this case.
In conclusion, as there is a significant increase in the CBG measurements when the finger is exposed to food, even if it was extensively wiped, patients should be advised to wash their hands with soap and water before performing the test. If this is not possible, the use of the second drop of blood is preferred to minimize the increment.
Acknowledgements
The authors acknowledge Bayer for their support, providing the strips for measuring glucose levels. They had no role in the study design, data collection, analysis, interpretation, or writing of this article. We have no other potential conflicts of interest relevant to this article.
References
- Klonoff DC, Blonde L, Cembrowski G, Chacra AR, Charpentier G,et al. (2011) Consensus report the current role of self-monitoring of blood glucose in non-insulin-treated type 2 diabetes. J Diabetes Sci Technol 5:1529-48.
- Self-Monitoring of Blood Glucose in Non-Insulin-Treated Type 2 Diabetes Recommendations based on a Workshop of the International Diabetes Federation Clinical Guidelines Taskforce in collaboration with the SMBG International Working Group.
- Guerci B, Drouin P, Grange V, Bougneres P, Fontaine P, et al. (2003)Self-monitoring of blood glucose significantly improves metabolic control in patients with type 2 diabetes mellitus: the Auto-Surveillance Intervention Active (ASIA) study Diabetes Metab29:587-94.
- Bergenstal R, Pearson J, Cembrowski GS, Bina D, Davidson J, et al. (2000) Identifying variables associated with inaccurate self-monitoring of blood glucose proposed guidelines to improve accuracy. Diabetes Educ. 26:981-9.
- American Diabetes Association(2015).
- Diabetes UK Self-blood glucose testing(2015).
- Diabetesvereniging Nederland Diabetes (2015).
- Hortensius J, Slingerland RJ, Kleefstra N, Logtenberg SJ, Groenier KH, et al. (2011) Self-monitoring of blood glucose the use of the first or the second drop of blood Diabetes Care 34:556-60.
- Arakawa M, Ebato C (2012) Influence of fruit juice on fingertips and patient behavior on self-monitoring of blood glucose Diabetes Res Clin Pract96: 50-2.
- Hirose T, Mita T, Fujitani Y, Kawamori R, Watada H (2011) Glucose monitoring after fruit peeling: pseudohyperglycemia when neglecting hand washing before fingertip blood sampling: wash your hands with tap water before you check blood glucose level. Diabetes Care34:596-7.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences